Lung Nodule Protocol Chest CT
Study Description
Computed tomography (CT) imaging is substantially more sensitive than chest X-ray for detection of lung nodules. A lung nodule protocol CT of the chest is further optimized to increase nodule conspicuity, improve accuracy of nodule measurement, and simultaneously obtain high resolution images of the lung parenchyma, all while minimizing radiation dose.
Wellstar Health System and the International Early Lung Cancer Action Program (I-ELCAP) have partnered to bring low DOSE-low COST screening lung imaging to the region. The goal is to increase long term survival of lung cancer through early detection. This investigational research study is aimed at detecting early stage lung cancer using low-dose computed tomography (CT) imaging.
Lung Cancer often displays no symptoms in an early stage and is thus rarely discovered, unless by accident. The late stage 5 year overall survival rate is less than 15%, and six out of ten people die within first year due to late stage discovery. However, when detected in early stage, the 10 year survival rate is 80%-92% based on I-ELCAP study published New England Journal of Medicine 2006.
Furthermore, the National Lung Screening Trial (NLST), a randomized national trial involving heavy smokers ages 55 to 74, compared the effects on lung cancer mortality of screening for lung cancer by low-dose helical computed tomography (CT) or by standard chest X-ray. Preliminary data reveals 20 percent fewer lung cancer deaths among trial participants screened with CT.
All I-ELCAP studies are done with low dose technique. This reduced visibility of mediastina and abdominal structures considerably, but still allows adequate visualization of bony structures and most parenchymal abnormalities. All non-contrast CT studies of the chest can be ordered with low dose technique (including pulmonary nodule protocol studies); this is particularly helpful when the patient is younger, or the abnormality is parenchymal or large (such as pleural effusions).
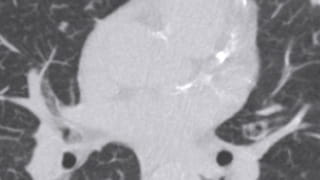
Patient Selection and Indications
For a lung nodule protocol CT scan:
- Known pulmonary nodule follow up
For the IELCAP trial:
- More than 40 years old and at least one of the following:
- Current smoker or prior smoking history.
- More than 10 year second-hand smoke exposure.
- Occupational exposure to cancer causing substances.
- And participants may not have had a chest CT within past year or any of the following symptoms or diagnosed lung disorders:
- Sarcoid Disease
- Idiopathic Pulmonary Fibrosis
- Interstitial Lung Disease
- Cough, coughing up blood, or unexplained weight loss.
Enrollment is easy, call (770) 956-STAR.
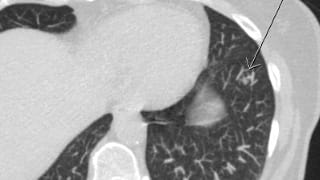
Patient Preparation
For a standard lung nodule protocol CT, no patient preparation is necessary. The low-dose CT involves no IV contrast or premedication.
For participation in the I-ELCAP trial, pre-registration with the I-ELCAP Program is necessary. Tell representative you wish to enroll in the Early Lung Cancer Screening Study. Screening CT is available at a reduced cost of $270.00 for research participants. The test is not typically covered by insurance, but a flexible spending account can be used. Cost may be tax deductible as a health care expense.
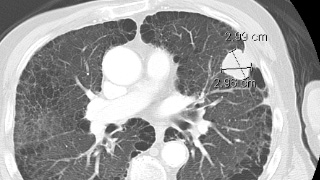
Reporting and Outcomes
Every lung nodule protocol CT exam is reviewed by a board-certified radiologist, and I-ECAP exams are only evaluated by subspecialty trained radiologists in the Cardiovascular imaging section of Quantum Radiology. A comprehensive report detailing all findings in the chest and in the imaged abdomen is provided to the referring clinician. If urgent findings are encountered, these are relayed in real time by telephone.
If the I-ELCAP scan is normal, it means no suspicious nodules were seen. A research participant is committed to return in 12 months for a follow up CT for baseline comparison. The follow up CT is also discounted.
Participants with any abnormal I-ELCAP scan will be evaluated by an affiliated Wellstar lung specialist for follow up. Follow up studies are usually considered diagnostic, and may be eligible for insurance coverage. Management depends upon variety of factors and is protocol guided.

